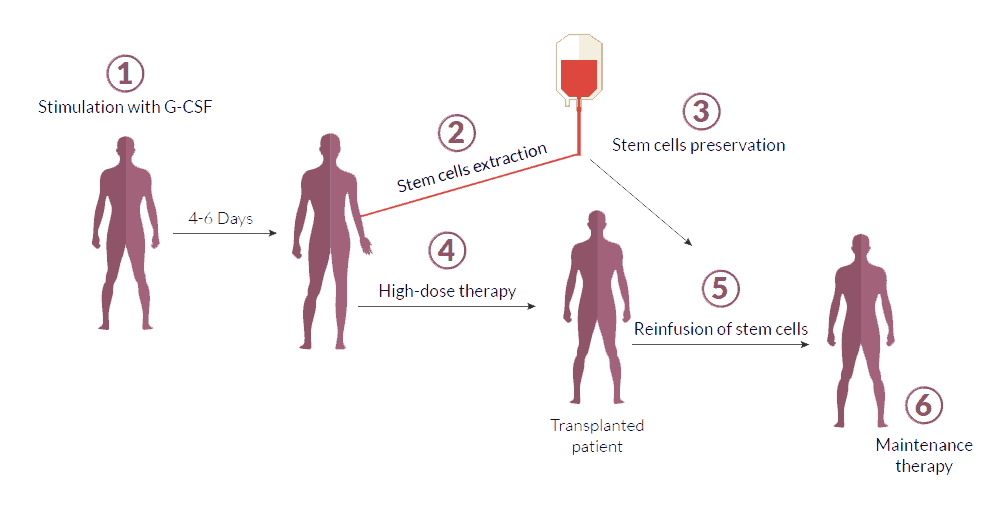
“These results are potentially practice changing because the drug is highly effective and easy to use,” said Eduard Schulz, M.D., Ph.D., of NIH’s Myeloid Malignancies Program, who was not involved in the trial. Results of the randomized phase 3 clinical trial, which was supported by NCI and BioLineRx, the company that makes motixafortide, were published April 17 in Nature Medicine. In the trial, a single injection of motixafortide given in addition to a series of injections of G-CSF, the drug most widely used to “mobilize” stem cells from the bone marrow to the blood, markedly increased the number of collected stem cells compared with treatment with G-CSF alone. The drug, motixafortide, helps blood-forming stem cells move from a person’s bone marrow into their bloodstream. New clinical trial results show that an experimental drug could help overcome this problem. But in about 15% to 30% of people who are fit enough for a transplant, the number of stem cells collected is not optimal for a successful transplant. 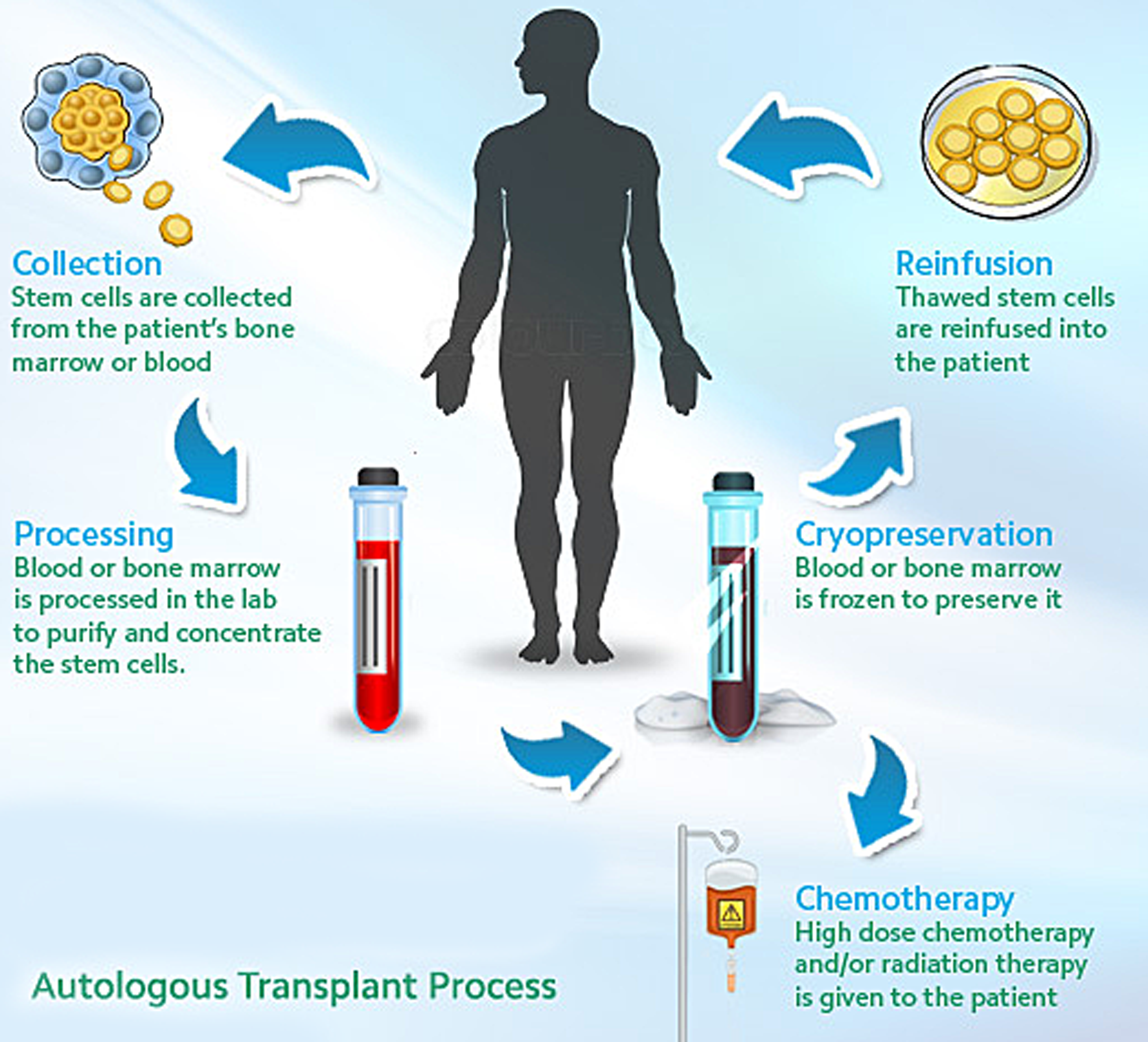
The approval, based on the results of the phase 3 GENESIS clinical trial, is for people with multiple myeloma who will undergo a transplant with their own stem cells, known as an autologous transplant.ĭetails of the GENESIS trial are discussed in the story below.įor people with multiple myeloma, treatment often includes high-dose chemotherapy to destroy cancerous blood cells, followed by a stem cell transplant using cells collected from the patient’s own blood to aid recovery. UPDATE: On September 8, 2023, the Food and Drug Administration approved motixafortide (Aphexda) for use with filgrastim (G-CSF) as part of preparing for a stem cell transplant.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
